Make your Christmas STEM challenge ideas last when you make these Christmas STEM fizzing ornaments using baking soda and vinegar chemical reactions. When it comes to mixing Christmas and science, you have to think outside the box a little bit like with these chemistry ornaments that can be part of any Christmas tree STEM activities.
My kids absolutely love making baking soda and vinegar eruptions, so I knew I wanted to incorporate it into our Christmas science experiments this season.

However, I didn’t want the experiment to just be a one and done sort of thing, so I tried to figure out a way to make the results last permanently.
Lucky for me (and you), this Christmas STEM fizzing ornaments project was a total success!
How to Make Baking Soda and Vinegar Paintings on Ornaments
You don’t need many ingredients to make your own fizzing paint Christmas science ornaments!
When you make erupting ornaments, the addition of the baking soda and vinegar painting (using acrylic paints), you can create really cool painted ornaments that last for years.
We first made our chemistry ornaments about 5 years ago, and they are still as good as new, so this method really works for creating long-lasting, science-themed ornaments for kids!
If you want a science activity that is a bit more STEAM-focused, then these chemistry christmas ornaments are perfect.
They are low-cost, easy-to-make, and are a fun way to add science into a craft activity!
Baking Soda and Vinegar Eruption Science
The science behind these ornaments is the classic chemical reaction between a base (the baking soda) and an acid (the vinegar).
When these two ingredients mix, they create a chemical reaction that creates sodium acetate, which then gives off a by-product of carbon dioxide.

The carbon dioxide is a gas that is released and causes the reaction to bubble. Since carbon dioxide is lighter than air, bubbles form!
The reaction between baking soda (sodium bicarbonate, NaHCO3) and vinegar (acetic acid, CH3COOH) is a classic example of an acid-base reaction.
In chemistry, substances are classified as acids or bases based on their properties. Acids taste sour, like lemon juice, while bases taste bitter, like soap. Vinegar is an acid, and baking soda is a base.
When you mix baking soda and vinegar together, something interesting happens. It fizzes and produces bubbles. This reaction is sometimes called a “chemical reaction.”
Books about Christmas Trees
These science books about evergreen trees will be a good addition to any Christmas tree STEM project!
Here’s what happens at the molecular level during this reaction:
- Baking soda (NaHCO3) is a base, and it contains sodium (Na+), hydrogen (H), carbon (C), and oxygen (O) atoms.
- Vinegar (CH3COOH) is an acid, and it contains carbon (C), hydrogen (H), and oxygen (O) atoms.
When you mix the baking soda (base) and vinegar (acid) together, they react because they want to balance each other out. This is like a chemical “tug of war.”
The reaction produces a new substance called carbon dioxide (CO2) gas. You see this gas escaping as bubbles, which is why you get the fizzing and bubbling when you mix them.
Along with the carbon dioxide gas, the reaction also forms water (H2O) and a substance called sodium acetate (NaCH3COO).
Books about Christmas Trees
These science books about evergreen trees will be a good addition to any Christmas tree STEM project!
This chemical reaction is like a balancing act because the acid and base are working together to neutralize each other. The result is the formation of new substances, including the gas, which you can see as bubbles.
When you add acrylic or tempera paint to these chemistry ornaments during this reaction, the acrylic paint bubbles and sticks to the sides of the ornaments, creating a unique design on the inside of the ornament.
It’s a fun way to mix art and science for the holiday season!
If you like these erupting ornaments, then you’ll also love the rest of these Christmas chemistry experiments.
Use our STEM extensions to scale this activity for preschool, elementary, or middle school!
What you’ll need for erupting Christmas ornaments:
Here is what you need to make your own Christmas STEM fizzing ornaments.

This post includes affiliate links for your convenience at no cost to you.
- Tempera paint
- Plastic funnels
- Baking soda
- Dish soap
- Vinegar
- Pipettes (we love these things and use them all the time!)
- Mason jars
- Large tray (we use this one all the time)
- Stirring rods
- Glitter
- Plastic ornaments with detachable tops
Hands-On STEM Resources about Trees
Here are some hands-on STEM resources about trees that can be used alongside this Christmas tree STEM activity.
Hands-On STEM Resources about Trees
Here are some hands-on STEM resources about trees that can be used alongside this Christmas tree STEM activity.
How to Paint Christmas Ornaments with a Chemical Reaction
I let the girls pick two colors of paint each, because I wasn’t sure how the colors would mix as the experiment progressed.
Bo chose silver and green and Monkey chose silver and red. They both also chose to add a sprinkle of glitter to their ornaments.

I set the ornaments upright in a mason jar and set them on the tray (this is to catch the drips).
The girls dropped the paint in (about a teaspoon of paint per ornament) and then I added a tablespoon of baking soda and a drop of dish soap on top of the paint.
The girls dipped their pipettes into the vinegar and gently squirted the baking soda with the vinegar.
They kept adding vinegar until the bubbles started to come out of the top.
I was afraid that too much vinegar would make all the paint leave the ornaments, but that didn’t seem to happen.
The girls didn’t flood the ornaments with vinegar, but they did put about four pipettes full of vinegar into each bottle.
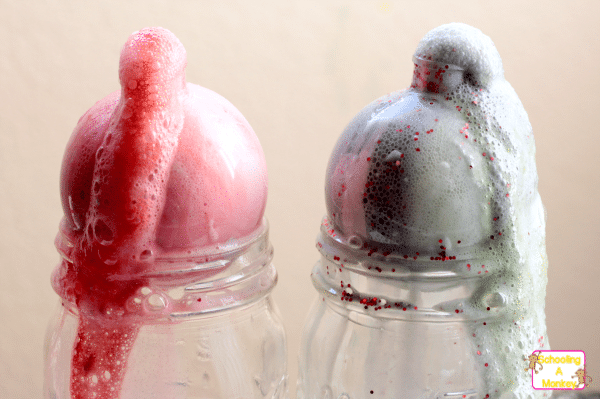
When the foaming slowed, I took the science ornaments over to the trash and dumped out the vinegar.
We then turned the ornaments upside down over the mason jars and set them in a window to dry.

The next day, the ornaments looked like this and were perfectly dry.
The girls were so pleased to see the results of their experiments in paint form.
Painted Fizzing Ornaments Variations
Here are some variations that you can try for different effects on your ornaments!
Baking Soda Snowflakes
- Make snowflake-shaped ornaments from baking soda and water (create a paste and shape it into snowflakes).
- Allow the snowflakes to dry completely.
- Decorate the dried snowflakes with markers or paint.
- Add a few drops of vinegar to make the snowflakes fizz and create a snowy effect.
Baking Soda Stenciled Ornaments
- Cut out stencils or use stickers to create designs on the surface of clear ornaments.
- Apply a thin layer of glue or Mod Podge over the stencil.
- Sprinkle baking soda over the glued area.
- Allow it to dry and shake off excess baking soda.
- When ready to decorate, add a few drops of vinegar to the stenciled area, causing a bubbly reaction.
STEM Extensions for Painted Fizzing Christmas Ornaments
STEM extensions for a lesson plan on baking soda and vinegar chemical reactions can help students explore related scientific concepts and engage in hands-on activities.
Here are some STEM extensions you can add to your lesson plan on fizzing Christmas ornaments:
Reaction Rate Experiment
Investigate how changing variables like temperature, concentration, or surface area affects the rate of the baking soda and vinegar reaction.
Collect data and create graphs to analyze the results.
Chemical Equations
Introduce chemical equations and the concept of stoichiometry by writing and balancing the equation for the baking soda and vinegar reaction (NaHCO3 + CH3COOH → CO2 + H2O + NaCH3COO).
Discuss the role of mole ratios in chemical reactions.
STEM Teaching Resources
These teaching resources will make your STEM classroom more fun and rewarding for your students!

Endothermic vs. Exothermic Reactions
Compare the baking soda and vinegar reaction to other chemical reactions.
Discuss the concepts of endothermic (absorbing heat) and exothermic (releasing heat) reactions.
Discover how temperature changes during the reaction.
Explore Acids and Bases
Connect the vinegar (acetic acid) to the concept of acids and discuss properties of acids and bases.
Test the pH levels of different liquids and classify them as acidic or basic.
Chemical Reaction Kinetics
Explore the concept of reaction kinetics by investigating the effect of catalysts (e.g., dish soap) on the reaction rate.
Discuss the role of activation energy in chemical reactions.
These STEM extensions can deepen students’ understanding of chemical reactions, acids and bases, reaction kinetics, and related scientific principles while fostering critical thinking and investigative skills.
Christmas STEM Fizzing Ornaments Lesson Plan
Here is a printable lesson plan you can use to go along with your STEAM activity when making painted fizzing ornaments!

How to Make Christmas Ornaments with a Chemical Reaction!
Get ready for a festive scientific adventure with this holiday-themed lesson plan for painted fizzing Christmas ornaments. Watch in amazement as baking soda and vinegar create erupting, fizzing ornaments—a perfect example of engaging chemical reactions. It's a must-try for holiday STEM!
Materials
- Tempera paint
- Plastic funnels
- Baking soda
- Dish soap
- Vinegar
- Pipettes (we love these things and use them all the time!)
- Mason jars
- Large tray (we use this one all the time)
- Stirring rods
- Glitter
- Plastic ornaments with detachable tops
Instructions
Here is a simple science lesson plan you can use while making fizzing Christmas ornaments.
Objective:
Students will create fizzing paint to decorate Christmas ornaments using a chemical reaction between baking soda and vinegar.
Students will observe and describe the chemical reaction.
Introduction (15 minutes):
Begin by discussing chemical reactions in simple terms, using the example of baking soda and vinegar creating bubbles.
Explain that students will be using this reaction to create fizzing paint for Christmas ornaments.
Activity (30 minutes):
Set out the clear Christmas ornaments, baking soda, vinegar, washable tempera paint in various colors, small funnels, pipettes or droppers, craft sticks or plastic spoons, and plastic cups.
a. Have students put on protective aprons or old t-shirts to avoid staining their clothes.
b. Instruct students to carefully remove the tops of the clear ornaments.
c. Using a small funnel, add about a teaspoon of baking soda into each ornament.
d. In plastic cups, mix the tempera paint with a small amount of vinegar until it becomes a liquid consistency. Explain that this mixture will react with the baking soda to create fizz.
e. Using pipettes or droppers, have students carefully add the paint-vinegar mixture to the ornaments. Encourage them to add different colors and experiment with patterns.
Observation and Discussion (10 minutes):
Athe paint-vinegar mixture reacts with the baking soda, students will see bubbles and fizzing inside the ornaments.
Discuss the chemical reaction they are witnessing and ask them to describe what they observe. Are there any changes in color or texture?
Conclusion (5 minutes):
Gather the students and discuss what they learned from the activity.
Ask questions such as:
What happened when we mixed baking soda and vinegar with paint?
How did the ornaments change during the reaction?
What did you enjoy most about this activity?
Recommended Products
As an Amazon Associate and member of other affiliate programs, I earn from qualifying purchases.
Teacher Christmas Tree Shirts
Here are some adorable Christmas tree shirts for teachers!
Chemist Tree Christmas Tree Teacher Shirt
Evergreen Christmas Tree Teacher Shirt
Math Christmas Tree Teacher Shirt
Books Christmas Tree Teacher Shirt
Pencil Christmas Tree Teacher Shirt

Forest Christmas Tree Teacher Shirt
Music Box Christmas Tree Teacher Shirt
Children Listen Christmas Tree Teacher Shirt
Music Note Christmas Tree Teacher Shirt
Teacher Christmas Tree Shirts
Here are some adorable Christmas tree shirts for teachers!
Chemist Tree Christmas Tree Teacher Shirt
Evergreen Christmas Tree Teacher Shirt
Math Christmas Tree Teacher Shirt
Books Christmas Tree Teacher Shirt
Pencil Christmas Tree Teacher Shirt

Forest Christmas Tree Teacher Shirt
Music Box Christmas Tree Teacher Shirt