There are a lot of crystal experiments out there! We love making all types of crystals. Today, we’re learning all about growing crystals with bluing and ammonia. It’s a fun twist on classic salt crystals and is one of our favorite classic science fair projects!

How to Grow Bluing and Ammonia Crystals
Follow along with these directions to learn how to grow bluing and ammonia crystals! This crystal garden science experiment is so beautiful and kids love doing it over and over!
Is There a Laundry Bluing Substitute?
This experiment is much, much easier if you use laundry bluing. However, you can’t always get it.
If you can’t find liquid laundry bluing, try your own DIY powdered laundry bluing:
How to Make Powdered Bluing
You can make your own bluing suspension by mixing three cups of baking soda with 1/2 a teaspoon of Prussian blue pigment powder. Mix this with distilled water in a 1 to 1 ratio to make your own homemade bluing.
How to Make Bluing Crystals a Science Fair Project
To make any science experiment or science demonstration a science fair project, all you need to add are variables to test!
You can use our bluing crystals lesson plan to transform your bluing and ammonia crystals into a complete science fair project or science experiment with a hypothesis, variables, and recording of data!
Bluing and Ammonia Crystal Hypothesis
For this crystal science experiment, all you need to start your experiment is a hypothesis!
Here are some variables to test:
- What happens if you don’t add bluing?
- Does ammonia make a difference in how fast the crystals form?
- Does the base material change how the crystals form?
If you want more crystal learning fun, check out how to grow salt crystals with salt and our list of classic science fair projects.
Bluing and Ammonia Crystal Science Explanation
Bluing and ammonia crystals are another type of salt crystal.
However, adding bluing changes the chemical composition of the crystals. Bluing contains a suspension of ferric ferrocyanide, which contains iron particles.
Instead of the salt crystals forming around each other like is typical in an ionic crystal, the crystals form a crystal “bloom” around the iron particles, which create much more organic-looking crystals that are delicate and look nothing like a standard square salt crystal.
The ammonia doesn’t change how the crystals form, but it does speed up the crystal formation process.

Supplies for the Bluing and Ammonia Crystal Experiment
Shop these Amazon affiliate links to pick up anything you’re missing before starting this experiment!
You’ll also need scissors, safety goggles, and kid-sized latex gloves for safety.
More Crystal Science Experiments
How to Make Instant Ice Crystals- Nucleation Science Experiment
How to Make Crystals without Borax
Borax Crystals Science Project
Crystal Science Lesson Plan Bundle
Directions for Growing Crystals with Bluing and Ammonia

Making bluing crystals is surprisingly easy! Here’s how to do it:
Mix 3 tablespoons of water, salt, and ammonia in a bowl.
Add 6 tablespoons of bluing to the same mixture and stir.
Cut your sponges into little squares. You can arrange them on a plate or put them in a jar like we did.
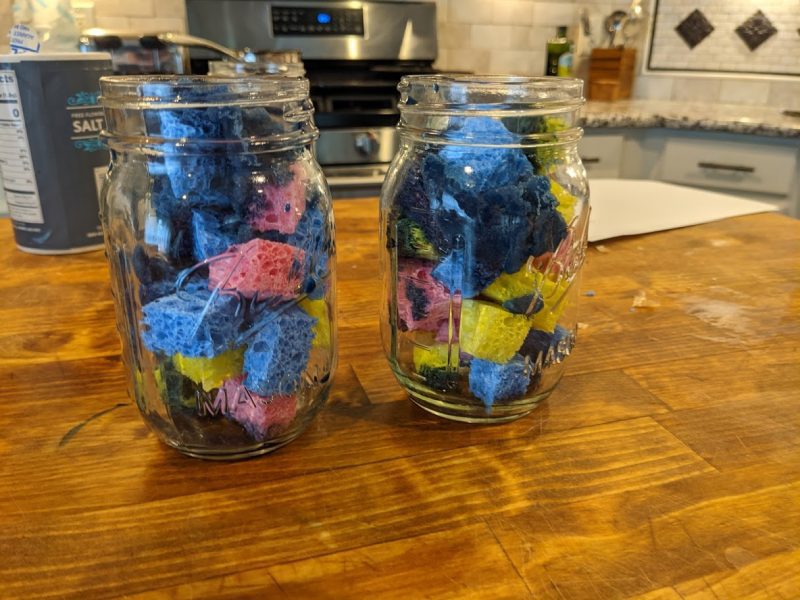
Pour the solution over the sponges.
Wait for several days.
You can add more liquid to the bottom of your container on day 2 or 3 if desired, but we found it wasn’t necessary.
After about 1-2 weeks, your feathery crystals will be ready for inspection!

Inspect these crystals with a magnifying glass to see how they look different from regular salt crystals.

