We love crystal experiments for any time of year. We love making seasonal versions of classic science experiments- especially ones that my kids love making, like crystals. For this St. Patrick’s Day, we decided to make salt crystal pots of gold to celebrate the pot of gold at the end of the rainbow! Follow along to learn how to grow crystals with salt with kids. It’s a fun twist on a classic and my kids were pleased at how they turned out.
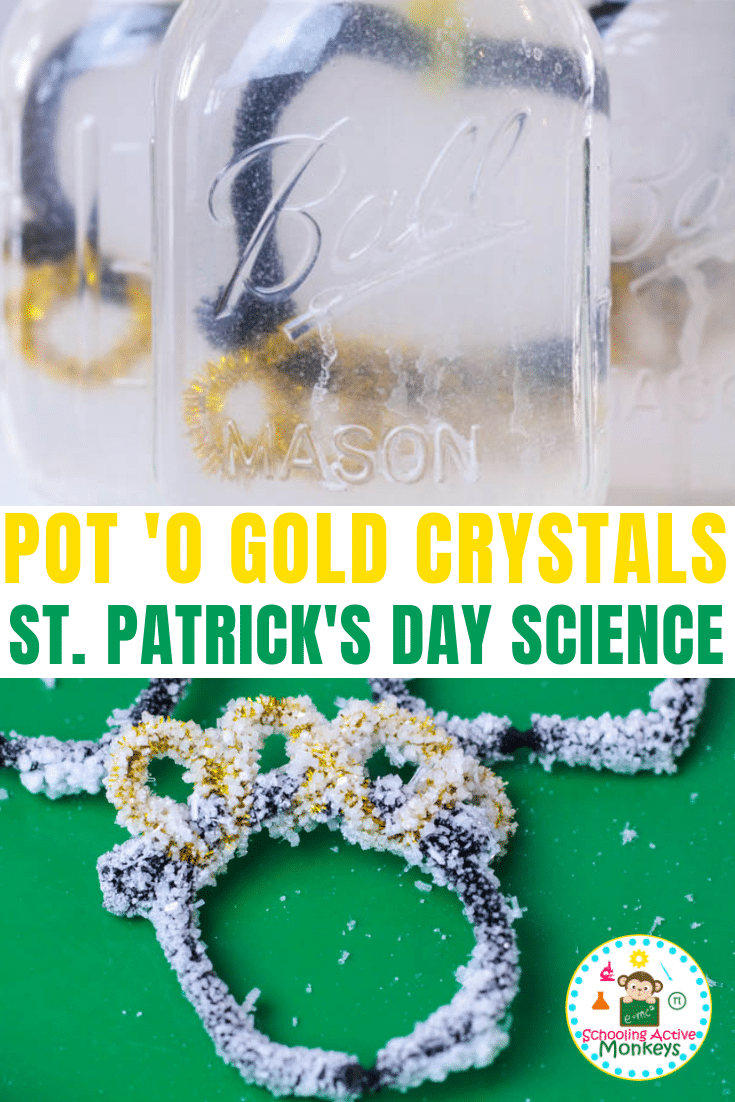
Keep reading to learn how to grow your own salt crystals for St. Patrick’s Day!
Table of Contents
How to Grow Crystals with Salt
Use this tutorial to learn how to grow crystals with salt in the shape of Pots of Gold for Saint Patrick’s Day!
Can you make crystals with normal salt?
You absolutely can make crystals with normal salt. Salt, in its natural state, is in a crystaline form.
The salt crystals are ground and purified to create table salt, but when you dissolve the salt particles, they will group together and turn back into large crystals!
You can also make Instant Ice Crystals, Bluing Crystals, Epsom Salt Crystals, or try the Borax Crystals Science Project.
St. Patrick’s Day Classroom Decorations
Here are some fun St. Patrick’s Day classroom decorations you can add to your classroom this March for festive flair and St. Paddy’s Day pride!
St Patrick’s Day Door Wreath for the Classroom
Pot of Gold Tinsel Tabletop Decoration
St. Patrick’s Day Rainbow Welcome Sign
St. Patrick’s Day Classroom Door Decorations
St. Patrick’s Day Window Clings for the Classroom
St. Patrick’s Day Door Decorations for the Classroom
Pot of Gold and Rainbow Ceiling Hanging
Lucky Hanging St. Patrick’s Decorations
St. Patrick’s Day Banner Decoration Set
Colorful St. Patrick’s Day Classroom Decoration Bundle
And check out even more St. Patrick’s Day teaching resources below!
- St. Patrick’s Teaching Resources
- St. Patrick’s Day Crafts
- St. Patrick’s Day Classroom Kits
- St. Patrick’s Classroom Decorations
Older kids will have a blast making a light up shamrock.
Of course, you won’t want to miss making rainbow slime for ST. Patrick’s Day! You can also try this glitter rainbow slime recipe or this fluffy rainbow slime recipe.
How do you grow salt crystals in a jar?
Growing salt crystals in a jar is as easy as forming salt crystals in any other fashion.
All you need is a string or some other flexible item (like a pipe cleaner) to suspend the object that you want to grow crystals on in the jar.
Use a glass jar and make sure the object doesn’t touch the sides or bottom of the jar.
How to Grow Salt Crystals Fast
What is the secret to creating crystals fast?
The secret is creating a super-saturated solution of salt water. A super-saturated solution is simply adding enough salt to the boiling water so that it is no longer able to dissolve.
How Do Salt Crystals Grow?
Salt crystals form when a solution of water becomes over-saturated with salt. When you create a super-saturated solution, the salt crystals must go somewhere when the mixture cools and the water starts to evaporate.
The furry pipe cleaners make the perfect surface for salt crystals to cling to. Over time, the crystals accumulate into larger and larger crystals.
As the water evaporates, the salt crystals cling to one another, forming large squares of salt, just like are found in nature.
Kids will be fascinated by the ionic bonds that salt makes, creating perfect squares and rectangles every time. The reason salt crystals make such perfect square shapes are due to the type of molecular bond that salt crystals have.
Teacher Apparel for St. Patrick’s Day
Here are some of the cutest shirts you can wear for St. Patrick’s Day at school!
Sequin Clover St. Patrick’s Day Teacher Shirt
Shenanigans Coordinator St. Patrick’s Day Teacher Shirt
My Students are Lucky Charms St. Patrick’s Day Teacher Shirt
Inclusive St Patrick’s Day Teacher Shirt
Lucky Clover St. Patrick’s Day Teacher Shirt
Have a Lucky Day Teacher Shirt for St. Patrick’s Day
Irish Alphabet St. Paddy’s Teacher Shirt
St. Patrick’s Day Shirt for Math Teachers
One Lucky Teacher St. Patrick’s Day Shirt
Salt bonds ionically, meaning that the bonds are inflexible. The molecules bond using the same pattern over and over, which creates a stable shape that is the same every time.
How long does it take to grow crystals with salt?
The time it takes to grow crystals with salt varies on how saturated your salt solution is.
The more saturated the solution, the faster the salt crystals will grow. With a supersaturated salt solution, you can see visible salt crystals within just an hour or two.
For solutions that are not supersaturated, it can take several days to see large salt crystals form.
St. Patrick’s Day Books for the Classroom
Use these books about St. Patrick’s Day to supplement your St. Patrick STEM activities this March!
St. Patrick’s Day STEM Activities
The Story Behind St. Patrick’s Day
Celebrations in My World: St. Patrick’s Day
Next Generation Science Standards (NGSS) Alignment
Use this experiment to complete the following NGSS standards:
- 5-PS1: Matter and Its Interactions
- 5-PS1-4: Conduct an investigation to determine whether the mixing of two or more substances results in new substances.
- PS1A: Structure and Properties of Matter
- PS1B: Chemical Reactions
Seasonal STEM Activities
One of my favorite parts of teaching is creating fun seasonal learning activities for my kiddos.

We particularly love doing STEM activities for kids and science experiments for kids. Today, we’re bringing a twist on how to grow salt crystals at home by learning how to make salt crystals!
We always like giving our science experiments a seasonal twist- and this time, we’re completing St. Patrick’s Day STEM activities by making pot ‘o gold salt crystals!
If you love this activity making St. Patrick’s Day pot ‘o gold salt crystals, then you’ll love how to make salt crystals without borax that makes an adorable salt crystal rainbow!
How to Conduct a Crystal Growing Science Experiment
Crystals of salt cover pots of gold with sodium chloride crystals at St. Patrick’s Day! Here’s how to complete the experiment:

This fun science activity shows how salt crystals form with a fun St. Patrick’s Day science twist.
How do you make homemade crystals?
There are just a few things necessary to make homemade crystals!
If you buy using these links, I will earn a commission on the sale.
The Mason Jar Scientist: 30 Jarring STEAM-Based ProjectsBall Glass Mason Jar with Lid and Band, Regular Mouth, 12 Jars
Acerich 300 Pcs Colored Pipe Cleaners Chenille Stems for DIY Art Craft Decorations (6 mm x 12 inch)
Morton Iodized Salt, 26 oz, Pack of 4
Ticonderoga Wood-Cased Graphite Pencils, #2 HB Soft, Yellow, 96 Count (13872)
AmazonBasics Pre-sharpened Wood Cased #2 HB Pencils, 150 Pack
More St. Patrick’s Day Activities for Kids
Try these hands on St. Patrick’s Day activities for kids!
- Super Fun and Easy Crystal Science Fair Projects
- St. Patrick’s Day STEAM Activities for Elementary
- St. Patrick’s Day STEM Sensory Bins
- Educational St. Patrick’s Day Activities
St. Patrick’s Day Salt Crystal Science Activity
Use this printable science activity to learn how to make these sparkling salt crystal pots of gold for Saint Patrick’s Day!
How to Make Salt Crystal Pots of Gold for Saint Patrick's Day

Follow along with these directions to learn how to make crystals from salt to create sparkling and shining pots of gold this St. Patrick's Day!
Materials
- Salt
- Pipe cleaners
- Pencils
- Mason jars
- Paper towels
Instructions
Boil enough water to fill as many jars as you have (we did 1 jar per pot of gold).
Add enough salt so that crystals form on the surface of the water. It will look like a sheet of thin ice.
Remove the water from the heat and let the mixture cool.
While the water is cooling, form your pots 'o gold from pipe cleaners. Use a black pipe cleaner to form the pot (we did a slightly squared circle).
Use a gold pipe cleaner to create the coins in the pot.
Attach the pots 'o gold to the pencils with another pipe cleaner.
Pour the saltwater into the jars.
Lower the pots 'o gold into the saltwater solution.
Wait a few hours for salt crystals to form. Leave the crystals in the solution for longer to make bigger crystals. After two hours, small salt crystals will start to form.
Pull the pots of gold out of the saltwater solution and lay on a paper towel until dry. 
Use a magnifying glass to study the salt crystals!
Notes
You can leave the pipe cleaners in the saltwater solution for 2-3 days, but if you are in a hurry, the crystals will be visible and ready to study within an hour or two.
Recommended Products
As an Amazon Associate and member of other affiliate programs, I earn from qualifying purchases.
St. Patrick STEM Activity Resources for the Classroom
Here are some fun St. Patrick’s themed STEM products you can use with your students this March!
St. Patrick’s Day STEM Activities
MagiCoders Rainbow Unicorn Coding Kit
St. Patrick’s Day Bingo Classroom Kit
Kaleidoscope STEAM Kit for the Classroom
Clixo Rainbow Engineering Classroom Set
Disappearing Rainbow Science Experiment